Reaction data
C3H5- + 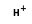 =
= 
Free energy of reaction at standard conditions (nominally 298.15 K, 1 atm.)
Go To: Top, References, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Data compiled by: John E. Bartmess
| ΔrG° (kJ/mol) | Method | Reference | Comment |
|---|---|---|---|
| 1672. ± 21. | H-TS | Peerboom, Rademaker, et al., 1992 | gas phase; B |
| 1671. ± 21. | H-TS | Graul and Squires, 1990 | gas phase; B |
| 1685. ± 8.8 | H-TS | DePuy, Gronert, et al., 1989 | gas phase; B |
| <1696.2 | N/A | Graul and Squires, 1988 | gas phase; B; Data excluded from overall average |
| >1653.5 | IMRB | Froelicher, Freiser, et al., 1986 | gas phase; B; Data excluded from overall average |
| >1653.5 | IMRB | Bohme, Lee-Ruff, et al., 1972 | gas phase; B; Data excluded from overall average |
References
Go To: Top, Free energy of reaction at standard conditions (nominally 298.15 K, 1 atm.), Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Peerboom, Rademaker, et al., 1992
Peerboom, R.A.L.; Rademaker, G.J.; Dekoning, L.J.; Nibbering, N.M.M.,
Stabilization of Cycloalkyl Carbanions in the Gas Phase,
Rapid Commun. Mass Spectrom., 1992, 6, 6, 394, https://doi.org/10.1002/rcm.1290060608
. [all data]
Graul and Squires, 1990
Graul, S.T.; Squires, R.R.,
Gas-Phase Acidities Derived from Threshold Energies for Activated Reactions,
J. Am. Chem. Soc., 1990, 112, 7, 2517, https://doi.org/10.1021/ja00163a007
. [all data]
DePuy, Gronert, et al., 1989
DePuy, C.H.; Gronert, S.; Barlow, S.E.; Bierbaum, V.M.; Damrauer, R.,
The Gas Phase Acidities of the Alkanes,
J. Am. Chem. Soc., 1989, 111, 6, 1968, https://doi.org/10.1021/ja00188a003
. [all data]
Graul and Squires, 1988
Graul, S.T.; Squires, R.R.,
On the Existence of Alkyl Carbanions in the Gas Phase,
J. Am. Chem. Soc., 1988, 110, 2, 607, https://doi.org/10.1021/ja00210a054
. [all data]
Froelicher, Freiser, et al., 1986
Froelicher, S.W.; Freiser, B.S.; Squires, R.R.,
The C3H5- isomers. Experimental and theoretical studies of the tautomeric propenyl ions and the cyclopropyl anion in the gas phase,
J. Am. Chem. Soc., 1986, 108, 2853. [all data]
Bohme, Lee-Ruff, et al., 1972
Bohme, D.K.; Lee-Ruff, E.; Young, L.B.,
Acidity order of selected bronsted acids in the gas phase at 300K,
J. Am. Chem. Soc., 1972, 94, 5153. [all data]
Notes
Go To: Top, Free energy of reaction at standard conditions (nominally 298.15 K, 1 atm.), References
- Symbols used in this document:
ΔrG° Free energy of reaction at standard conditions - Data from NIST Standard Reference Database 69: NIST Chemistry WebBook
- The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. However, NIST makes no warranties to that effect, and NIST shall not be liable for any damage that may result from errors or omissions in the Database.
- Customer support for NIST Standard Reference Data products.