Reaction data
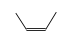 =
= 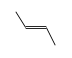
Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.)
Go To: Top, References, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Data compiled by: Hussein Y. Afeefy, Joel F. Liebman, and Stephen E. Stein
| ΔrH° (kJ/mol) | Method | Reference | Comment |
|---|---|---|---|
| -4.34 ± 0.04 | Eqk | Kapteijn, Steen, et al., 1983 | gas phase; Between 250-823 K; ALS |
| -4.18 ± 0.08 | Eqk | Akimoto, Sprung, et al., 1972 | gas phase; ALS |
| -4.18 | Eqk | Meyer and Stroz, 1972 | gas phase; At 300 K; ALS |
| -2.7 ± 0.4 | Eqk | Happel, Hnatow, et al., 1971 | gas phase; ALS |
| -1.8 | Eqk | Maccoll and Ross, 1965 | gas phase; GC; ALS |
| -5.0 ± 0.4 | Eqk | Golden, Egger, et al., 1964 | gas phase; ALS |
| -3. | Ciso | Levanova and Andreevskii, 1964 | gas phase; At 420.3 K; ALS |
References
Go To: Top, Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.), Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Kapteijn, Steen, et al., 1983
Kapteijn, F.; Steen, A.J.; Mol, J.C.,
Thermodynamics of the geometrical isomerization of 2-butene and 2-pentene,
J. Chem. Thermodyn., 1983, 15, 137-146. [all data]
Akimoto, Sprung, et al., 1972
Akimoto, H.; Sprung, J.L.; Pitts, J.N., Jr.,
Nitrogen dioxide catalyzed geometric isomerization of 2-butene and 2-pentene. A precise method for determining the enthalpies and entropies of geometric isomerizations,
J. Am. Chem. Soc., 1972, 94, 4850-4855. [all data]
Meyer and Stroz, 1972
Meyer, E.F.; Stroz, D.G.,
Thermodynamics of n-butene isomerization,
J. Am. Chem. Soc., 1972, 94, 6344-6347. [all data]
Happel, Hnatow, et al., 1971
Happel, J.; Hnatow, M.A.; Mezaki, R.,
Isomerization equilibrium constants of n-butenes,
J. Chem. Eng. Data, 1971, 16, 206-209. [all data]
Maccoll and Ross, 1965
Maccoll, A.; Ross, R.A.,
The hydrogen bromide catalyzed isomerization of n-butenes. I. equilibrium values,
J. Am. Chem. Soc., 1965, 87, 1169-1170. [all data]
Golden, Egger, et al., 1964
Golden, D.M.; Egger, K.W.; Benson, S.W.,
Iodine-catalyzed isomerization of olefins. I. Thermodynamics data from equilibrium studies of positional and geometrical isomerization of 1-butene and 2-butene,
J. Am. Chem. Soc., 1964, 86, 5416-5420. [all data]
Levanova and Andreevskii, 1964
Levanova, S.V.; Andreevskii, D.N.,
The equilibrium of 2-chlorobutane dehydrochlorination,
Neftekhimiya, 1964, 4, 329-336. [all data]
Notes
Go To: Top, Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.), References
- Symbols used in this document:
ΔrH° Enthalpy of reaction at standard conditions - Data from NIST Standard Reference Database 69: NIST Chemistry WebBook
- The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. However, NIST makes no warranties to that effect, and NIST shall not be liable for any damage that may result from errors or omissions in the Database.
- Customer support for NIST Standard Reference Data products.