Trimethyl tin, positive ion
Reaction thermochemistry data
Go To: Top, Ion clustering data, References, Notes
Data compilation copyright
by the U.S. Secretary of Commerce on behalf of the U.S.A.
All rights reserved.
Data compiled by: Michael M. Meot-Ner (Mautner) and Sharon G. Lias
Note: Please consider using the
reaction search for this species. This page allows searching
of all reactions involving this species. A general reaction search
form is
also available. Future versions of this site may rely on
reaction search pages in place of the enumerated reaction
displays seen below.
Individual Reactions
C3H9Sn+ + 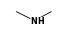 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H7N = (C3H9Sn+ • C2H7N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 185. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| ΔrH° | 185. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 127. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 118. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| 115. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 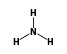 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + H3N = (C3H9Sn+ • H3N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 154. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 122. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 90.4 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
C3H9Sn+ + 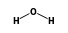 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + H2O = (C3H9Sn+ • H2O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 108. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 115. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 46.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
C3H9Sn+ + 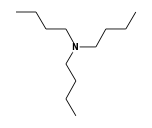 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C12H27N = (C3H9Sn+ • C12H27N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 201. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 144. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 126. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 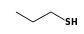 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H8S = (C3H9Sn+ • C3H8S)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 143. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 135. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 72.4 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O2 = (C3H9Sn+ • C5H10O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 175. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 101. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 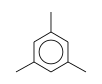 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C9H12 = (C3H9Sn+ • C9H12)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 134. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 64.0 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 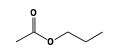 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O2 = (C3H9Sn+ • C5H10O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 168. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 137. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 96.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 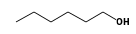 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H14O = (C3H9Sn+ • C6H14O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 157. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 139. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 83.7 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 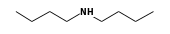 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C8H19N = (C3H9Sn+ • C8H19N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 204. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 141. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 130. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 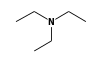 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H15N = (C3H9Sn+ • C6H15N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 191. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 142. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 117. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 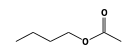 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H12O2 = (C3H9Sn+ • C6H12O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 174. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 101. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 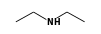 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H11N = (C3H9Sn+ • C4H11N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 194. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 122. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 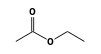 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H8O2 = (C3H9Sn+ • C4H8O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 168. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 95.8 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 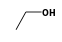 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H6O = (C3H9Sn+ • C2H6O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 146. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 135. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 74.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H10O = (C3H9Sn+ • C4H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 153. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 81.6 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 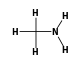 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + CH5N = (C3H9Sn+ • CH5N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 176. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 128. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 109. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H3N = (C3H9Sn+ • C2H3N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 157. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 131. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 87.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 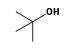 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H10O = (C3H9Sn+ • C4H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 153. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 82.0 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 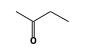 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H8O = (C3H9Sn+ • C4H8O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 164. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 137. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 92.5 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 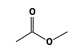 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H6O2 = (C3H9Sn+ • C3H6O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 161. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 89.1 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 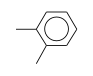 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C8H10 = (C3H9Sn+ • C8H10)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 125. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 55.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 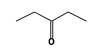 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O = (C3H9Sn+ • C5H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 165. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 132. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 95.8 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 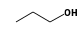 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H8O = (C3H9Sn+ • C3H8O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 149. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 130. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 78.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 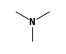 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H9N = (C3H9Sn+ • C3H9N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 191. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 130. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 120. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 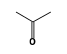 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H6O = (C3H9Sn+ • C3H6O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 156. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 129. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrG° | 88.7 | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ + 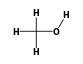 = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + CH4O = (C3H9Sn+ • CH4O)
Ion clustering data
Go To: Top, Reaction thermochemistry data, References, Notes
Data compilation copyright
by the U.S. Secretary of Commerce on behalf of the U.S.A.
All rights reserved.
Data compiled by: Michael M. Meot-Ner (Mautner) and Sharon G. Lias
Note: Please consider using the
reaction search for this species. This page allows searching
of all reactions involving this species. Searches may be limited
to ion clustering reactions. A general reaction search form is
also available.
Clustering reactions
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + CH4O = (C3H9Sn+ • CH4O)
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + CH5N = (C3H9Sn+ • CH5N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 176. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 128. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 109. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H3N = (C3H9Sn+ • C2H3N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 157. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 131. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 87.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H6O = (C3H9Sn+ • C2H6O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 146. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 135. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 74.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C2H7N = (C3H9Sn+ • C2H7N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 185. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| ΔrH° | 185. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 127. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 118. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| 115. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H6O = (C3H9Sn+ • C3H6O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 156. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 129. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrG° | 88.7 | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H6O2 = (C3H9Sn+ • C3H6O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 161. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 89.1 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H8O = (C3H9Sn+ • C3H8O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 149. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 130. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 78.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H8S = (C3H9Sn+ • C3H8S)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 143. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 135. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 72.4 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C3H9N = (C3H9Sn+ • C3H9N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 191. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 130. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 120. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H8O = (C3H9Sn+ • C4H8O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 164. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 137. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 92.5 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H8O2 = (C3H9Sn+ • C4H8O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 168. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 95.8 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H10O = (C3H9Sn+ • C4H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 153. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 81.6 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H10O = (C3H9Sn+ • C4H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 153. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 136. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 82.0 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C4H11N = (C3H9Sn+ • C4H11N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 194. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 122. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O = (C3H9Sn+ • C5H10O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 165. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 132. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 95.8 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O2 = (C3H9Sn+ • C5H10O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 175. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 101. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C5H10O2 = (C3H9Sn+ • C5H10O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 168. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 137. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 96.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H12O2 = (C3H9Sn+ • C6H12O2)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 174. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 140. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 101. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H14O = (C3H9Sn+ • C6H14O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 157. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 139. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 83.7 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C6H15N = (C3H9Sn+ • C6H15N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 191. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 142. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 117. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C8H10 = (C3H9Sn+ • C8H10)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 125. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 55.2 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C8H19N = (C3H9Sn+ • C8H19N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 204. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 141. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 130. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C9H12 = (C3H9Sn+ • C9H12)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 134. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 133. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 64.0 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + C12H27N = (C3H9Sn+ • C12H27N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 201. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 144. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 126. | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH, Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + H2O = (C3H9Sn+ • H2O)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 108. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 115. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 46.9 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH; Entropy change calculated or estimated |
C3H9Sn+ +  = (C3H9Sn+ •
= (C3H9Sn+ •  )
)
By formula: C3H9Sn+ + H3N = (C3H9Sn+ • H3N)
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrH° | 154. | kJ/mol | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
| Quantity |
Value |
Units |
Method |
Reference |
Comment |
| ΔrS° | 122. | J/mol*K | N/A | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
Free energy of reaction
| ΔrG° (kJ/mol) |
T (K) |
Method |
Reference |
Comment |
| 90.4 | 525. | PHPMS | Stone and Splinter, 1984 | gas phase; switching reaction((CH3)3Sn+)CH3OH/NH3, Entropy change calculated or estimated |
References
Go To: Top, Reaction thermochemistry data, Ion clustering data, Notes
Data compilation copyright
by the U.S. Secretary of Commerce on behalf of the U.S.A.
All rights reserved.
Stone and Splinter, 1984
Stone, J.A.; Splinter, D.E.,
A high-pressure mass spectrometric study of the binding of (CH3)3Sn+ to lewis bases in the gas phase,
Int. J. Mass Spectrom. Ion Processes, 1984, 59, 169. [all data]
Notes
Go To: Top, Reaction thermochemistry data, Ion clustering data, References
- Symbols used in this document:
| T |
Temperature |
| ΔrG° |
Free energy of reaction at standard conditions |
| ΔrH° |
Enthalpy of reaction at standard conditions |
| ΔrS° |
Entropy of reaction at standard conditions |
- Data from NIST Standard Reference Database 69:
NIST Chemistry WebBook
-
The National Institute of Standards and Technology (NIST)
uses its best efforts to deliver a high quality copy of the
Database and to verify that the data contained therein have
been selected on the basis of sound scientific judgment.
However, NIST makes no warranties to that effect, and NIST
shall not be liable for any damage that may result from
errors or omissions in the Database.
-
Customer support
for NIST Standard Reference Data products.