Reaction data
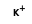 +
+ 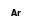 = (
= (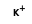 •
• 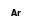 )
)
Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.)
Go To: Top, References, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Data compiled as indicated in comments:
RCD - Robert C. Dunbar
M - Michael M. Meot-Ner (Mautner) and Sharon G. Lias
| ΔrH° (kJ/mol) | Method | Reference | Comment |
|---|---|---|---|
| 14. ± 7.1 | CIDT | Rodgers and Armentrout, 2000 | RCD |
| 8.24 | IMob | Gatland, 1984 | gas phase; M |
| 12.0 | SCATTERING | Gislason, 1984 | gas phase; M |
| 12.3 | IMob | Viehland, 1984 | gas phase; M |
| 11.5 | IMob | Takebe, 1983 | gas phase; M |
| 11.0 | IMob | Robson and Kumar, 1973 | gas phase; M |
| 13.2 | IMob | Skullerud, 1973 | gas phase; M |
| 11.7 | IMob | Mason and Sharp, 1958 | gas phase; M |
| 12. | IMob | Mason and Sharp, 1958 | gas phase; M |
References
Go To: Top, Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.), Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Rodgers and Armentrout, 2000
Rodgers, M.T.; Armentrout, P.B.,
Noncovalent Metal-Ligand Bond Energies as Studied by Threshold Collision-Induced Dissociation,
Mass Spectrom. Rev., 2000, 19, 4, 215, https://doi.org/10.1002/1098-2787(200007)19:4<215::AID-MAS2>3.0.CO;2-X
. [all data]
Gatland, 1984
Gatland, I.R.,
Swarms of Ions and Electrons in Gases, W. Lindinger, T. D. Mark and F. Howorka, eds. (Springer, New York, 1984, 1984, 44. [all data]
Gislason, 1984
Gislason, E.A.,
Quoted in I. R. Gatland in Swarms of Ions and Electrons in Gases, W. Lindinger, T. D. Mark and F. Howorka, eds. (Springer, New York, 1984, 1984, 44. [all data]
Viehland, 1984
Viehland, L.A.,
Interaction Potentials for Li+ - Rare - Gas Systems,
Chem. Phys., 1984, 78, 2, 279, https://doi.org/10.1016/0301-0104(83)85114-3
. [all data]
Takebe, 1983
Takebe, M.,
The Generalized Mobility Curve for Alkali Ions in Rare Gases: Clustering Reactions and Mobility Curves,
J. Chem. Phys., 1983, 78, 12, 7223, https://doi.org/10.1063/1.444763
. [all data]
Robson and Kumar, 1973
Robson, R.E.; Kumar, K.,
Mobility and Diffusion II. Dependence on Experimental Variables and Interaction Potential for Alkali Ions in Rare Gases,
Aust. J. Phys., 1973, 26, 2, 187, https://doi.org/10.1071/PH730187
. [all data]
Skullerud, 1973
Skullerud, H.R.,
Mobility, Diffusion and Interaction Potential for Potassium Ions in Argon,
J. Phys. B., 1973, 6, 5, 918, https://doi.org/10.1088/0022-3700/6/5/027
. [all data]
Mason and Sharp, 1958
Mason, E.A.; Sharp, H.W.,
Mobility of gaseous lons in weak electric fields,
Ann. Phys., 1958, 4, 3, 233, https://doi.org/10.1016/0003-4916(58)90049-6
. [all data]
Notes
Go To: Top, Enthalpy of reaction at standard conditions (nominally 298.15 K, 1 atm.), References
- Symbols used in this document:
ΔrH° Enthalpy of reaction at standard conditions - Data from NIST Standard Reference Database 69: NIST Chemistry WebBook
- The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. However, NIST makes no warranties to that effect, and NIST shall not be liable for any damage that may result from errors or omissions in the Database.
- Customer support for NIST Standard Reference Data products.