Reaction data
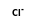 +
+ 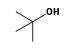 = (
= (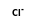 •
•  )
)
Reaction thermochemistry data
Go To: Top, References, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Data compiled as indicated in comments:
B - John E. Bartmess
M - Michael M. Meot-Ner (Mautner) and Sharon G. Lias
| Quantity | Value | Units | Method | Reference | Comment |
|---|---|---|---|---|---|
| ΔrH° | 77. ± 20. | kJ/mol | AVG | N/A | Average of 6 values; Individual data points |
| Quantity | Value | Units | Method | Reference | Comment |
| ΔrS° | 115. | J/mol*K | PHPMS | Hiraoka and Mizuse, 1987 | gas phase; M |
| ΔrS° | 100. | J/mol*K | PHPMS | Sieck, 1985 | gas phase; M |
| ΔrS° | 97.9 | J/mol*K | N/A | Larson and McMahon, 1984 | gas phase; Entropy change calculated or estimated; French, Ikuta, et al., 1982; M |
| ΔrS° | 110. | J/mol*K | PHPMS | Kebarle, 1977 | gas phase; M |
| ΔrS° | 43.1 | J/mol*K | N/A | Yamdagni and Kebarle, 1971 | gas phase; Entropy change calculated or estimated; M |
| Quantity | Value | Units | Method | Reference | Comment |
| ΔrG° | 48.45 | kJ/mol | TDAs | Bogdanov, Peschke, et al., 1999 | gas phase; B |
| ΔrG° | 51.5 ± 1.3 | kJ/mol | TDAs | Sieck, 1985 | gas phase; B |
| ΔrG° | 48.53 | kJ/mol | TDAs | Hiraoka and Mizuse, 1987 | gas phase; B |
| ΔrG° | 46.4 ± 8.4 | kJ/mol | IMRE | Larson and McMahon, 1984, 2 | gas phase; B,M |
| ΔrG° | 46.4 ± 8.4 | kJ/mol | TDAs | Yamdagni and Kebarle, 1971 | gas phase; B |
References
Go To: Top, Reaction thermochemistry data, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Hiraoka and Mizuse, 1987
Hiraoka, K.; Mizuse, S.,
Gas-Phase Solvation of Cl- with H2O, CH3OH, C2H4OH, i-C3H7OH, n-C3H7OH, and t-C4H9OH,
Chem. Phys., 1987, 118, 3, 457, https://doi.org/10.1016/0301-0104(87)85078-4
. [all data]
Sieck, 1985
Sieck, L.W.,
Thermochemistry of Solvation of NO2- and C6H5NO2- by Polar Molecules in the Vapor Phase. Comparison with Cl- and Variation with Ligand Structure.,
J. Phys. Chem., 1985, 89, 25, 5552, https://doi.org/10.1021/j100271a049
. [all data]
Larson and McMahon, 1984
Larson, J.W.; McMahon, T.B.,
Gas phase negative ion chemistry of alkylchloroformates,
Can. J. Chem., 1984, 62, 675. [all data]
French, Ikuta, et al., 1982
French, M.A.; Ikuta, S.; Kebarle, P.,
Hydrogen bonding of O-H and C-H hydrogen donors to Cl-. Results from mass spectrometric measurement of the ion-molecule equilibria RH + Cl- = RHCl-,
Can. J. Chem., 1982, 60, 1907. [all data]
Kebarle, 1977
Kebarle, P.,
Ion Thermochemistry and Solvation from Gas Phase Ion Equilibria,
Ann. Rev. Phys. Chem., 1977, 28, 1, 445, https://doi.org/10.1146/annurev.pc.28.100177.002305
. [all data]
Yamdagni and Kebarle, 1971
Yamdagni, R.; Kebarle, P.,
Hydrogen bonding energies to negative ions from gas phase measurements of ionic equilibria,
J. Am. Chem. Soc., 1971, 93, 7139. [all data]
Bogdanov, Peschke, et al., 1999
Bogdanov, B.; Peschke, M.; Tonner, D.S.; Szulejko, J.E.; McMahon, T.B.,
Stepwise solvation of halides by alcohol molecules in the gas phase,
Int. J. Mass Spectrom., 1999, 187, 707-725, https://doi.org/10.1016/S1387-3806(98)14180-5
. [all data]
Larson and McMahon, 1984, 2
Larson, J.W.; McMahon, T.B.,
Hydrogen bonding in gas phase anions. An experimental investigation of the interaction between chloride ion and bronsted acids from ICR chloride exchange equilibria,
J. Am. Chem. Soc., 1984, 106, 517. [all data]
Notes
Go To: Top, Reaction thermochemistry data, References
- Symbols used in this document:
ΔrG° Free energy of reaction at standard conditions ΔrH° Enthalpy of reaction at standard conditions ΔrS° Entropy of reaction at standard conditions - Data from NIST Standard Reference Database 69: NIST Chemistry WebBook
- The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. However, NIST makes no warranties to that effect, and NIST shall not be liable for any damage that may result from errors or omissions in the Database.
- Customer support for NIST Standard Reference Data products.