Reaction data
(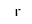 •
• 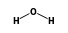 ) +
) +  = (
= (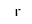 • 2
• 2 )
)
- Reaction by formula: (I- • H2O) + H2O = (I- • 2H2O)
- Comments:
- Bond type: Hydrogen bond (negative ion to hydride)
- Information on this page:
- Options:
Reaction thermochemistry data
Go To: Top, References, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Data compiled as indicated in comments:
B - John E. Bartmess
M - Michael M. Meot-Ner (Mautner) and Sharon G. Lias
| Quantity | Value | Units | Method | Reference | Comment |
|---|---|---|---|---|---|
| ΔrH° | 41.4 ± 0.84 | kJ/mol | TDAs | Keesee and Castleman, 1980 | gas phase; B,M |
| ΔrH° | 39.7 | kJ/mol | N/A | Markovich, Pollack, et al., 1994 | gas phase; For photodissociation plus electron loss. Affinity is difference from lower solvated ion.; B |
| ΔrH° | 39.7 ± 0.84 | kJ/mol | TDAs | Hiraoka, Mizuse, et al., 1988 | gas phase; B,M |
| ΔrH° | 41. ± 4.2 | kJ/mol | TDAs | Payzant, Yamdagni, et al., 1971 | gas phase; B |
| ΔrH° | 41. ± 4.2 | kJ/mol | TDAs | Arshadi, Yamdagni, et al., 1970 | gas phase; B,M |
| Quantity | Value | Units | Method | Reference | Comment |
| ΔrS° | 73.6 | J/mol*K | PHPMS | Hiraoka, Mizuse, et al., 1988 | gas phase; M |
| ΔrS° | 84.9 | J/mol*K | HPMS | Keesee and Castleman, 1980 | gas phase; M |
| ΔrS° | 79.5 | J/mol*K | HPMS | Arshadi, Yamdagni, et al., 1970 | gas phase; M |
| Quantity | Value | Units | Method | Reference | Comment |
| ΔrG° | 17. ± 2. | kJ/mol | AVG | N/A | Average of 6 values; Individual data points |
Free energy of reaction
| ΔrG° (kJ/mol) | T (K) | Method | Reference | Comment |
|---|---|---|---|---|
| 17. | 300. | HPMS | Banic and Iribarne, 1985 | gas phase; electric fields; M |
References
Go To: Top, Reaction thermochemistry data, Notes
Data compilation copyright by the U.S. Secretary of Commerce on behalf of the U.S.A. All rights reserved.
Keesee and Castleman, 1980
Keesee, R.G.; Castleman, A.W., Jr.,
Gas phase studies of hydration complexes of Cl- and I- and comparison to electrostatic calculations in the gas phase,
Chem. Phys. Lett., 1980, 74, 139. [all data]
Markovich, Pollack, et al., 1994
Markovich, G.; Pollack, S.; Giniger, R.; Cheshnovsky, O.,
Photoelectron spectroscopy of Cl-, Br-, and I- solvated in water clusters,
J. Chem. Phys., 1994, 101, 11, 9344, https://doi.org/10.1063/1.467965
. [all data]
Hiraoka, Mizuse, et al., 1988
Hiraoka, K.; Mizuse, S.; Yamabe, S.,
Solvation of Halide Ions with H2O and CH3CN in the Gas Phase,
J. Phys. Chem., 1988, 92, 13, 3943, https://doi.org/10.1021/j100324a051
. [all data]
Payzant, Yamdagni, et al., 1971
Payzant, J.D.; Yamdagni, R.; Kebarle, P.,
Hydration of CN-, NO2-, NO3-, and HO- in the gas phase,
Can. J. Chem., 1971, 49, 3308. [all data]
Arshadi, Yamdagni, et al., 1970
Arshadi, M.; Yamdagni, R.; Kebarle, P.,
Hydration of Halide Negative Ions in the Gas Phase. II. Comparison of Hydration Energies for the Alkali Positive and Halide Negative Ions,
J. Phys. Chem., 1970, 74, 7, 1475, https://doi.org/10.1021/j100702a014
. [all data]
Banic and Iribarne, 1985
Banic, C.M.; Iribarne, J.V.,
Equilibrium Constants for Clustering of Neutral Molecules about Gaseous Ions,
J. Chem. Phys., 1985, 83, 12, 6432, https://doi.org/10.1063/1.449543
. [all data]
Notes
Go To: Top, Reaction thermochemistry data, References
- Symbols used in this document:
T Temperature ΔrG° Free energy of reaction at standard conditions ΔrH° Enthalpy of reaction at standard conditions ΔrS° Entropy of reaction at standard conditions - Data from NIST Standard Reference Database 69: NIST Chemistry WebBook
- The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. However, NIST makes no warranties to that effect, and NIST shall not be liable for any damage that may result from errors or omissions in the Database.
- Customer support for NIST Standard Reference Data products.